Rethinking Primate Use in Diabetes Research: The Rise of Human-Relevant NAMs is ready!
- Oct 28, 2025
- 6 min read
While cynomolgus macaques have historically contributed to some preclinical studies, the assertion that they are essential for advancing diabetes treatments is no longer tenable. Human-relevant New Approach Methodologies — including iPSC-derived islet organoids, organ-on-chip microphysiological systems, encapsulated stem-cell β-cell therapies now in human trials, and advanced in-silico PK/PD models — provide more predictive, ethically preferable, and regulatorily-accepted routes to development. Regulators (FDA, EMA) and funders (NC3Rs, NIH, EU programs) are actively supporting these methods, and recent clinical progress with stem-cell derived islets demonstrates direct translation in humans without exclusive dependence on non-human primates. Nature, US FDA

Reason & Necessity for this Article
Mauritius - in it's quest to promote the island as a pre-clinical research hub and the continued use of the local long tailed macacques (cynomolgus macaques) in biomedical research - has recently invited various international biomedical researchers to speak in favour of using primates in biomedical research at regional conferences, press releases and video publications.
Dr Melanie Graham, Director of the Preclinical Research Centre at the University of Minnesota, USA, a specialist in translational medicine, who reportedly works on cutting-edge therapies for diabetes - a disease that affects nearly one in five adults in Mauritius - in an interview of a video series by the Mauritian Cyno Breeders Association and Defimedia group titled 'mots de recherche' highlighted " the essential and often overlooked role of Mauritian cynomolgus macaques in the development of innovative therapies: stem cell transplantation, insulin-producing cell replacement, ongoing clinical trials, and more.... These advances have been made possible thanks to these preclinical models, which are bringing medicine closer to a sustainable treatment... and even, one day, a real cure."
➡️ Graham's statements and the efforts of the pro-primate experimentation conglomerate to influence public opinion
require analysis and rectification.
Short verdict
Cynomolgus macaques have historically been useful in some translational diabetes studies, but the claim that they are essential and irreplaceable is no longer accurate.
A rapidly maturing toolbox of human-relevant New Approach Methodologies (NAMs) — human iPSC/islet organoids, organ-on-chip microphysiological systems, encapsulated stem-cell β-cell therapies (now in human trials), advanced in-silico models (PBPK and immune models), and improved clinical approaches (microdosing, adaptive early-phase designs) — already replace many primate uses and are being accepted by regulators. The scientific and regulatory landscape is actively shifting in favor of these alternatives. Nature US FDA
Where macaques have helped (but why that doesn’t make them irreplaceable)
Past role: Non-human primates (NHPs) have been used for some immunology, transplantation, and chronic physiology studies because of anatomical and immunological similarities to humans. That made them attractive for a narrow class of experiments (e.g., long-term graft survival, large-animal device/implant tolerability).
Why this is not decisive today: The primary justifications (closer physiology, size, long lifespan) are being addressed by human-derived systems that give more directly relevant human biology, by engineered encapsulation and immunomodulation solutions already in clinical testing, and by advanced computational methods that predict PK/PD and immune interactions. In other words: NHPs can model some aspects, but they are not the only or necessarily the best route to reliable human translation anymore. Nature PubMed
NAMs and breakthroughs that replace primate use in diabetes research
1) Human induced-pluripotent-stem-cell (iPSC)-derived islets and islet organoids
What they do: generate glucose-responsive, insulin-producing β-cells and multicellular islet organoids from human cells for disease modelling, toxicity testing, and cell-replacement strategies.Why it replaces primates: these are human cells that recapitulate pancreatic islet physiology and human-specific responses — far more directly predictive than cross-species extrapolation. They are used for mechanistic studies, drug screening, and even as the source material for clinical cell therapies.Key refs: reviews and 2024–25 literature on islet organoids and clinical translation. PMC1 PMC2
2) Encapsulated / implantable stem cell-derived β-cell therapies (clinical evidence)
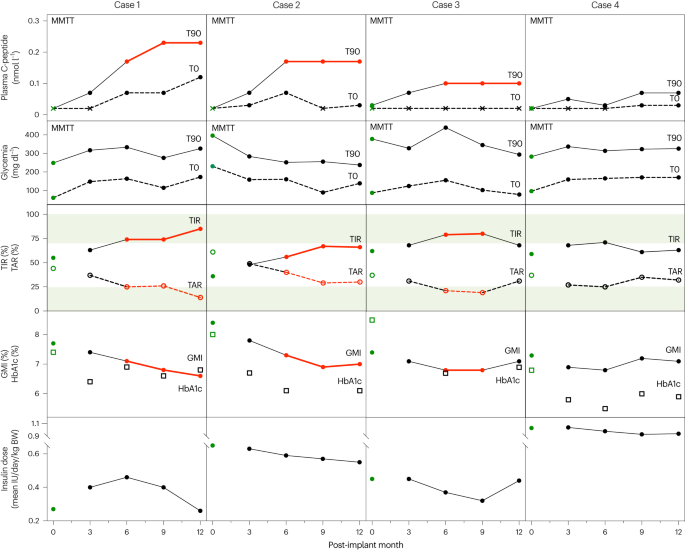
What they do: deliver stem cell-derived insulin-producing cells inside semi-permeable devices or via direct infusion into liver/pancreas; some designs avoid lifelong immunosuppression (encapsulation) and are now in human trials.Why it replaces primates: these therapies are already in humans (early to mid clinical trials), which is the strongest evidence that preclinical reliance on primates is not strictly necessary to advance to human testing. Their development has depended heavily on human cell manufacturing and in-vitro functional testing, not primate prediction alone.Key refs: Vertex (VX-880 / zimislecel) and ViaCyte programs; Nature report on encapsulated β cells. Vrtx Nature
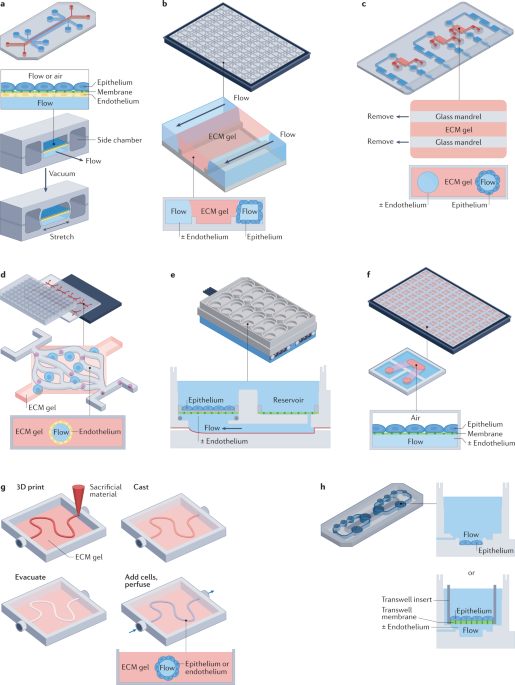
3) Pancreas / islet microphysiological systems (organ-on-chip)
What they do: microfluidic devices recreate islet microenvironment (oxygen, perfusion, vascular and immune interactions) enabling continuous physiologic readouts (insulin release, viability) and drug testing.Why it replaces primates: MPS allow human-specific PK/PD and mechanistic toxicity studies at organ-level resolution and can be linked with liver/immune chips to test systemic effects — a direct substitute for many systemic studies previously done in NHPs.Key ref: a 2024 pancreas-on-chip microphysiological system and authoritative reviews on organs-on-chips. RSC Publishing Nature
4) Advanced computational models and PBPK / QSP (Quantitative Systems Pharmacology)
What they do: integrate human in vitro data (metabolism, transporter activity), clinical biomarker data and physiology to predict human PK/PD and dose-response without relying on animal scaling.Why it replaces primates: For many drugs (especially biologics with known human targets), PBPK & QSP coupled to human cell data can predict human exposures and target engagement more accurately than interspecies scaling from macaques. Regulators are explicitly encouraging these methods. U.S. Food and Drug Administration US FDA
5) Human immune-system-based assays (cytokine-release, co-cultures, 3D immune tissues)
What they do: ex vivo human blood assays, iPSC-derived immune cells and 3D co-cultures test immunogenicity, cytokine release and immune-cell interactions.Why it replaces primates: Human immune assays detect human-specific immune activation (e.g., lessons learned after TGN1412). Using human blood/cell assays before first-in-human dosing is now standard and far more predictive than NHP immunology in some contexts. Nature
6) Clinical-first strategies: microdosing, adaptive early trials, and human challenge paradigms
What they do: give carefully calculated microdoses or adaptive small cohorts with intensive human biomarker readouts, reducing the need for extensive animal bridging.Why it replaces primates: For well-characterized classes or therapies with strong human in vitro evidence, regulators now accept human-first approaches supported by NAMs for safety evaluation. (FDA/EMA roadmaps and pilot programs are enabling this.) U.S. Food and Drug Administration EMA
Concrete examples showing the shift from NHPs to NAMs (evidence)
Concrete New Approach Methodologies (NAMs) and other human-relevant advances that can and already do replace or greatly reduce the role of cynomolgus macaques:
Encapsulated stem cell β cells: positive interim human outcomes published and cited as a major translational step (Nature 2023; human trials ongoing). This is direct human evidence that bypassed exclusive dependence on primate data. Nature
Vertex VX-880 (zimislecel): stem cell–derived islet cell therapy in Phase 1/2 and moving to pivotal studies; clinical efficacy signals have been reported (reduced insulin needs). This shows human trials are progressing on human-derived cell products. ClinicalTrials
Organs-on-chip & MPS: multiple groups have published pancreas/islet chips and argue these devices can model human islet physiology for drug screening and mechanistic studies. Regulators and funders (NC3Rs, NIH, EU/EMA) are investing heavily in these platforms. RSC Publishing NC3Rs
Regulatory & institutional momentum away from routine primate use
FDA & EMA are actively endorsing NAMs and have produced roadmaps to reduce animal testing in preclinical safety packages; pilot programs encourage submissions that rely on validated NAMs. This is not just advocacy — it’s regulatory change. U.S. Food and Drug Administration EMA
Funding and validation programs (NC3Rs, NIH, EU) are prioritizing organ-on-chip, MPS, and human-derived models to accelerate adoption and regulatory acceptance. NC3Rs 1 NC3Rs 2

Final thoughts for Mauritius
With the NAMs and breakthroughs that already replace primate use in diabetes research and the strong trend away from animal models in all fields of biomedical research Mauritius can no longer fall for the old narrative of indispencability of use of primates in research.
The public and policy makers have the right to be informed on the true facts on todays use of primates in research.
Policy makers should take note of this and fulfil their duty to lead Mauritius into a future that combines progress, sustainable development and economic growth with ethical modern research.
A call for investment in local NAM capacity
Instead allowing and promoting the use of primates in research we ask that local NAM capacity be developed (e.g., iPSC labs, organ chip hubs, partnerships with clinical cell-therapy centers) so Mauritius can lead with human-relevant research and transition primate facilities to alternative, sustainable biotech.
Note of acknowledgment:
While there are still some minor hurdles before a complete shift to NAMs — i.e.
a) some complex whole-body physiology and long-term outcomes (e.g., chronic immunology, device biocompatibility over years) can at this time still pose challenges for NAMs - i.e. some device and surgical testing will still rely on large animal models for now — but not necessarily primates (pigs and other non-primate large animals remain options for device/tissue mechanics) and
b) Regulatory inertia: although regulators are updating frameworks, sponsors still face variability in expectations for novel agents; companies often use conservative animal packages for global submissions —
The trend, however, is strongly toward fewer animals and more human-relevant data.
The shift to full repacement of animal models with NAMs is already on the way. Clining to outdated research methods to protect the continued exploitation of the Mauritius primates is unethical - both towards the animals and towards human patients - as NAMs offer more reliable results.
Reference Articles :





Comments